- Home
- Countries & Regions
- Asia
- Indonesia
- Topics & Events
- Workshop of Pharmacovigilance Inspection Guideline
Topics & Events
February 18, 2022
Workshop of Pharmacovigilance Inspection Guideline
Japan International Cooperation Agency (JICA) in collaboration with Direktorat Pengawasan Keamanan, Mutu, dan Ekspor Impor Obat, Narkotika, Psikotropika, Prekursor dan Zat Adiktif (Ditwas. KMEI ONPPZA), Badan POM held workshop of Pharmacovigilance Inspection Guideline on 17th -18th of February 2022. The workshop aims to strengthen the competencies of Badan POM and its Provincial Offices (Unit Pelaksana Teknis/UPT BPOM) in implementing pharmacovigilance (PV) inspection to pharmaceutical industries in Indonesia.
Badan POM has a mandate to ensure that circulating drugs in Indonesia meet the requirements for safety, efficacy and quality. Therefore, Badan POM conducts supervision from the pre-market to the post-market stage by implementing PV system to detect, assess, elucidate and prevent adverse effects and other medicine-related problems.
In supporting PV implementation in Indonesia, JICA and Ditwas. KMEI ONPPZA developed PV Inspection Guideline, as one of the PV activities, to support Badan POM and UPT BPOM in PV inspections to fulfil their responsibilities for good pharmacovigilance practices and risk management system.
Four PV inspectors from Indonesia and one expert from Japan were brought as source person to guide participants to understand more about the guideline. Dra. Mayagustina Andarini, Apt, M.Sc, Deputy Chairperson for Drug, Narcotics, Psychotropics, Precursors, and Addictive Substances Control of Badan POM highlighted that those pharmaceutical industries should be encouraged to implement pharmacovigilance appropriately in good management. On this occasion, Mr. Sano Yoshihiko, Chief Advisor of JICA-BPOM Project shared the knowledge about the implementation of PV system in Japan, including PV inspection that was conducted in Osaka Prefecture.
Dra. Tri Asti Isnariani, Apt, M.Pharm, Dra. Ega Febrina, Apt, Ms Megrina Dian Agustin, S.Si, Apt, and Dra. Fachdiana Fidia, Apt, M.Si also explained more deeply about PV regulation and inspection for Pharmaceutical Industries in Indonesia, summary of PV system (SPS) and tools for PV inspection. Furthermore, simulation in working groups were facilitated to provide a real and concrete picture of what PV inspectors have to do during the inspection.
Approximately 80 participants in total from Badan POM and UPT BPOM in Jakarta, Bandung, Serang, Semarang, Surabaya, Yogyakarta, Padang, Medan, Palembang, Bogor, Tangerang, Kediri and Surakarta participated in this workshop.
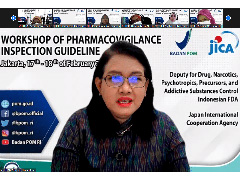 Dra. Mayagustina Andarini, Apt, M.Sc.
Dra. Mayagustina Andarini, Apt, M.Sc.
 Mr. Sano Yoshihiko
Mr. Sano Yoshihiko
 Dra. Tri Asti Isnariani, Apt, M.Pharm
Dra. Tri Asti Isnariani, Apt, M.Pharm
 (Clockwise) Dra. Ega Febrina, Apt; Ms Megrina Dian Agustin, S.Si, Apt; and dr. Rahmaniah, M.Biomed.
(Clockwise) Dra. Ega Febrina, Apt; Ms Megrina Dian Agustin, S.Si, Apt; and dr. Rahmaniah, M.Biomed.
 (Clockwise) Dra. Fachdiana Fidia, Apt, M.Si; Miyanto, S.Farm, Apt; and one participant from Balai.
(Clockwise) Dra. Fachdiana Fidia, Apt, M.Si; Miyanto, S.Farm, Apt; and one participant from Balai.
 Photo group session between all speakers and participants
Photo group session between all speakers and participants

- Asia
- Southeast Asia
- Cambodia
- Indonesia
- Laos
- Malaysia
- Myanmar
- Philippines
- Thailand
- Timor-Leste
- Viet Nam
- East Asia
- China
- Mongolia
- Central Asia and the Caucasus
- Armenia
- Azerbaijan
- Georgia
- Kyrgyz Republic
- Tajikistan
- Uzbekistan
- South Asia
- Afghanistan
- Bangladesh
- Bhutan
- India
- Maldives
- Nepal
- Pakistan
- Sri Lanka
- Oceania
- Latin America
- Africa
- Middle East
- Europe
- Asia
- About JICA
- News & Features
- Countries & Regions
- Our Work
- Thematic Issues
- Types of Assistance
- Partnerships with Other Development Partners
- Climate Change / Environmental and Social Considerations
- Evaluations
- Compliance and Anti-corruption
- Science and Technology Cooperation on Global Issues
- Research
- JICA Development Studies Program / JICA Chair
- Support for the Acceptance of Foreign HRs / Multicultural and Inclusive Community
- Publications
- Investor Relations
